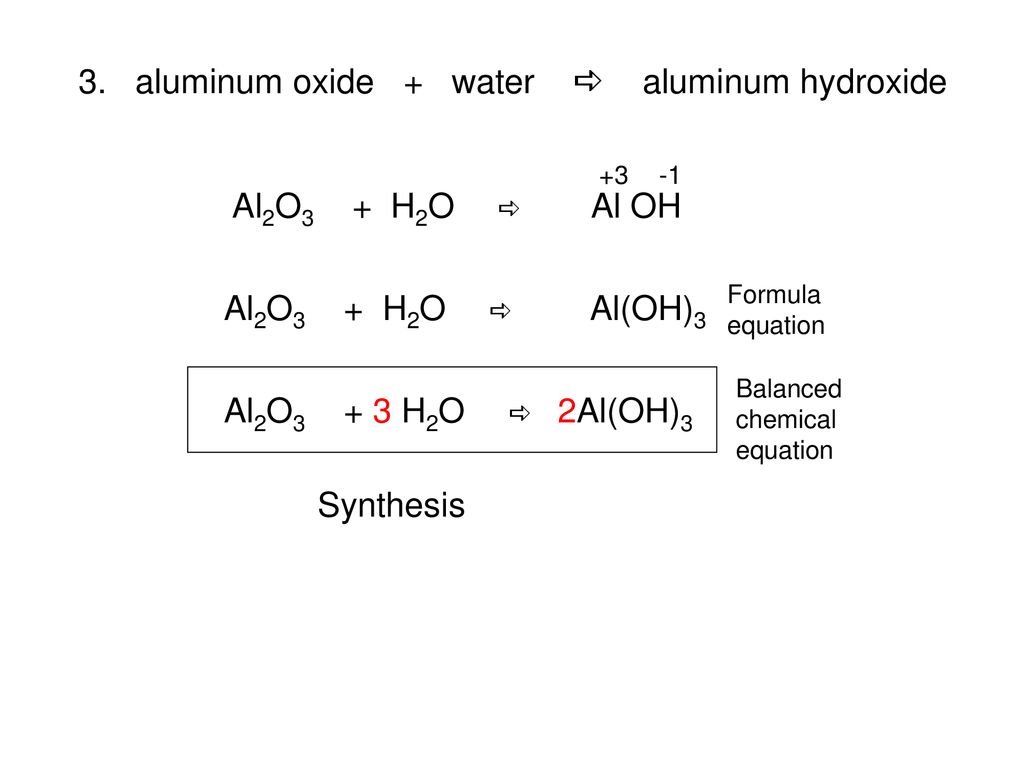
Question: Balance following equations: C3H8 O2 → CO2 H2O Al2(SO3)3 NaOH → Na2SO3 Al(OH)3 Al2O3 - Chem 101 - Stuvia US

High-Pressure δ-Al(OH)3 and δ-AlOOH Phases and Isostructural Hydroxides/Oxyhydroxides: New Structural Insights from High-Resolution 1H and 27Al NMR | The Journal of Physical Chemistry B

0.078 g Al(OH)(3). is dehydrated to Al(2)O(3). The Al(2)O(3) so obtained reacted with 6 milli-equivalent of HCl. The equivalent of AlCl(3) produced during the reaction are:

High-Pressure δ-Al(OH)3 and δ-AlOOH Phases and Isostructural Hydroxides/Oxyhydroxides: New Structural Insights from High-Resolution 1H and 27Al NMR | The Journal of Physical Chemistry B

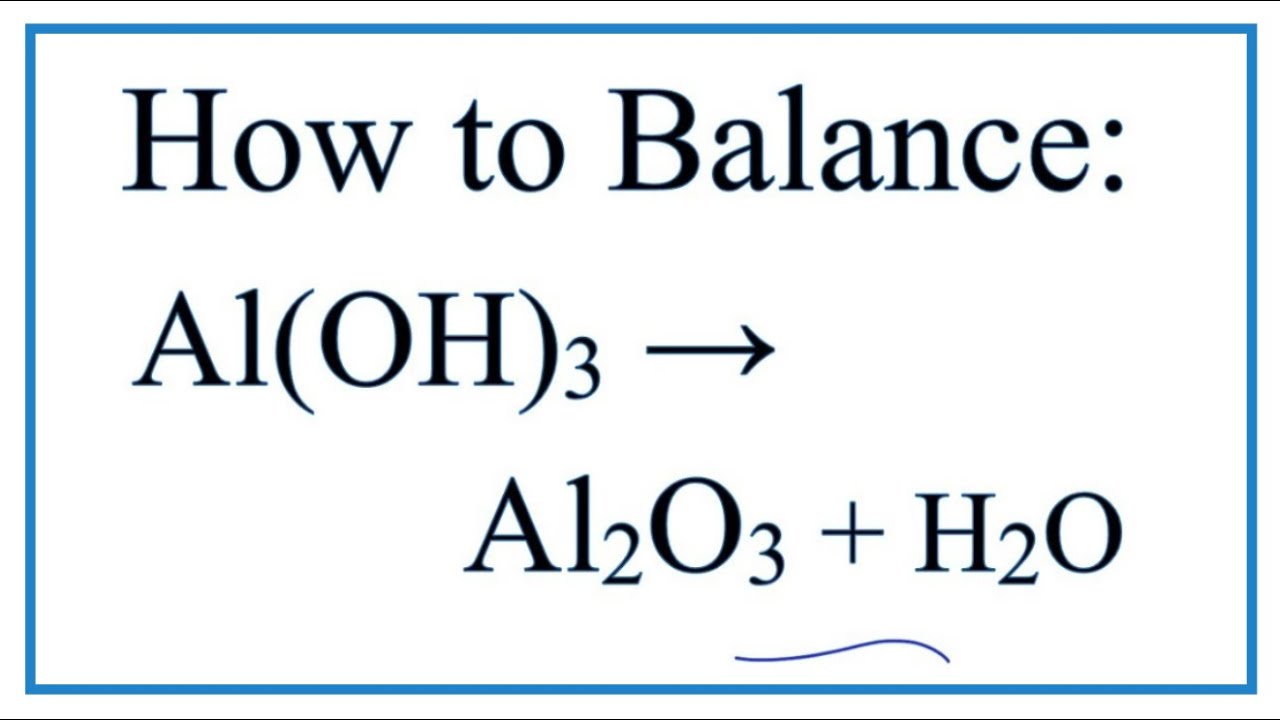
Balance the following equation and identify what type of reaction it is. Al( OH)3 arrow Al2O3 + H2O | Homework.Study.com